ISO 31010:2019 Risk Management – Foundation Course
This ISO 31010 Foundation Risk Management Course is one of deGRANDSON’s three ISO 31010 courses and is designed to provide learners with basic knowledge of risk assessment techniques and their general application.
ISO 31010 and FDA Guidance Document Q9(R1) Quality Risk Management
On 03-May-23, the Food and Drug Administration (FDA) announced the publication of a final guidance for the industry entitled “Q9(R1) Quality Risk Management.” The guidance was prepared under the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH).
The revision is intended to provide guidance on quality risk management principles and tools that can be applied to different aspects of pharmaceutical quality. Annex I: Quality Risk Management Methods and Tools lists the recommended tools and methods. These are…
- Basic Risk Management Facilitation Methods
- Failure Mode Effects Analysis (FMEA)
- Failure Mode, Effects, and Criticality Analysis (FMECA)
- Fault Tree Analysis (FTA)
- Hazard Analysis and Critical Control Points (HACCP)
- Hazard Operability Analysis (HAZOP)
- Preliminary Hazard Analysis (PHA)
- Risk Ranking and Filtering
All of these tools and methods are included in our Foundation Course.
Key Features:
- Course is accredited to ISO 21001, the standard for Educational Organizations Management Systems
- An ISO 31010 Risk Management certification is awarded upon passing the certification exam
- Certificate is immediately available online after the successful passing of the exam
- Certificate comes with a shareable QR code for instant verification of credentials
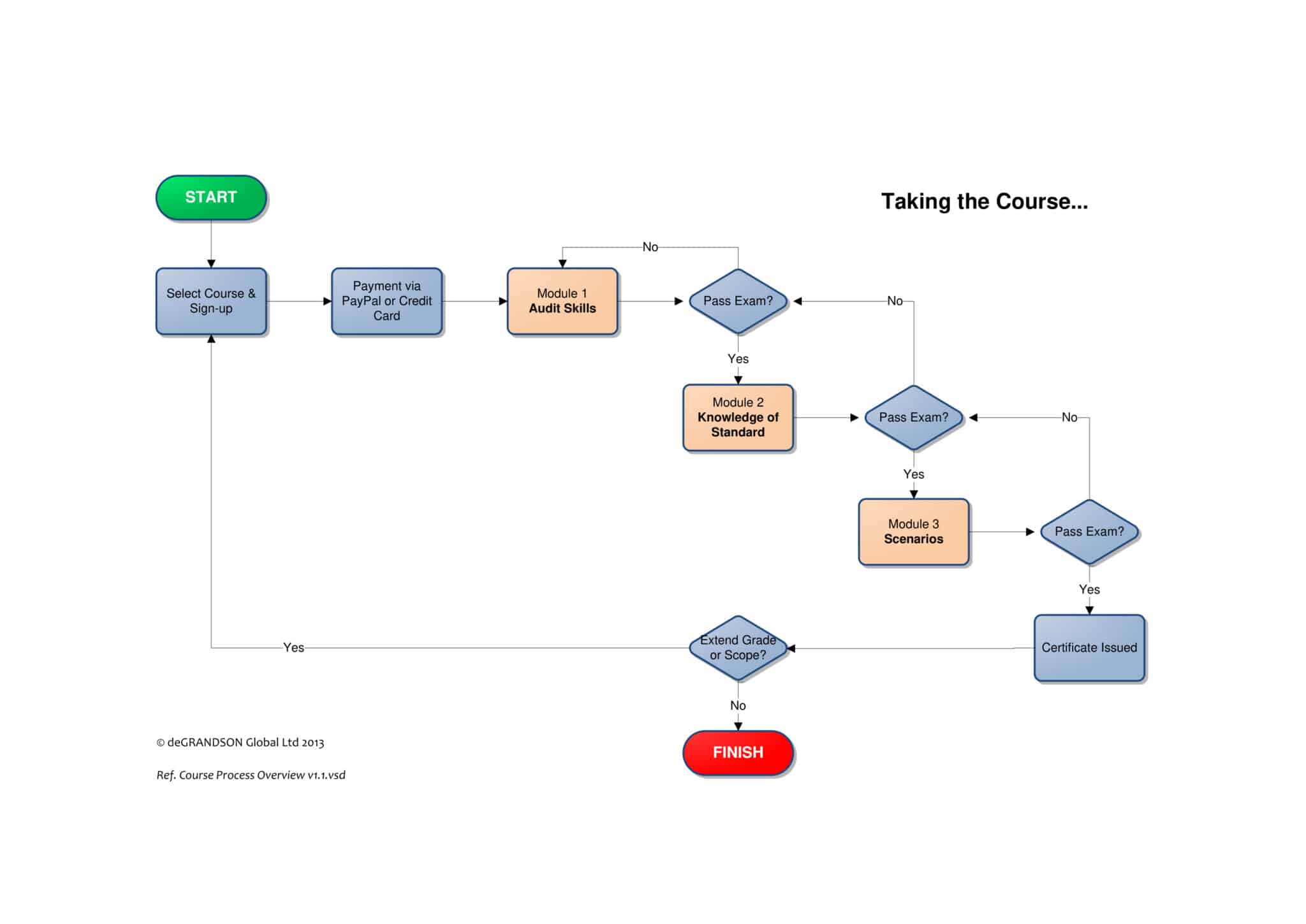
- Lessons range from 15 minutes to 1 hour, typically 20–30 minutes, ensuring that each topic is covered in suitable detail
- Course includes practice with scenarios that include dialogues
- Course includes 24/7 Live-chat Learner Support
- Course includes a learner manual, a copy of the standard, and samples of relevant forms and other documents
- Course comes with full-audio narration and Closed Captions for accessibility
- Courses are hosted on your browser so that no software has to be downloaded avoiding security risks.
- Course comes with quizzes, practice with scenarios, and open-book certification exam
- Course is hosted on your browser so that no software has to be downloaded avoiding security risks.
- Features cross-device compatibility (courses can be taken on any desktop, tablet, or mobile)
- Offers full-resume feature (end a session mid-lesson and continue exactly where you left off, even from a different device)
- Features real-time interactive content in a secure web-based environment
- Offers a clear learning path (once you've completed the internal auditor course, you have the option to progress to the lead auditor, up to the consultant and lead auditor course.
- Examination and certificate fee are already included in the course fee
- Payable via PayPal or Stripe using any credit or debit cards
- Option to pay in 4 monthly installments available
Who should enroll in this ISO 31010 Risk Management Foundation Course?
This course is intended for:
- anyone involved in assessing or managing risk;
- people who are involved in developing guidance that sets out how risk is to be assessed in specific contexts;
- people who need to make decisions where there is uncertainty including:
- those who commission or evaluate risk assessments,
- those who need to understand the outcomes of assessments, and
- those who have to choose assessment techniques to meet particular needs.
What will I learn from this ISO 31010 Risk Management Foundation Course?
This comprehensive Course covering all aspects of medical device risk management is divided into four Modules …
- Module 0: Introduction and background to the Course - Gain an understanding of the foundations of risk management, its importance in organizational decision-making, and how ISO 31010 and ISO 31000 work together to support effective risk assessment and control.
- Module 1: Introduction to Risk Management, includes …
-
ISO 31010 – Knowledge and Application — Learn the purpose and scope of ISO 31010, focusing on its role in providing risk assessment techniques that support ISO 31000’s broader risk management framework.
-
Terms & Definitions — Understand essential terminology used in risk management, including definitions related to risk, uncertainty, controls, and residual risk.
-
Risk Management Principles — Explore the guiding principles of effective risk management, emphasizing value creation, integration into organizational processes, and continuous improvement.
-
Risk Management Framework — Learn how to establish a structured framework for risk management aligned with ISO 31000, ensuring clear governance, accountability, and communication pathways.
-
Risk Management Process Overview — Review the universal steps of risk management, including context establishment, risk assessment, risk treatment, monitoring, and communication.
-
- Module 2: The Risk Management Process (ISO 31000)
-
The Universal Risk Management Process Model — Study the systematic model of ISO 31000, including the key phases of risk identification, analysis, evaluation, and treatment, and how these apply across diverse industries and operational environments.
-
- Module 3: Risk Assessment Techniques: Foundation (ISO-IEC 31010), including …
-
Brainstorming — Learn how to use brainstorming sessions to identify potential risks collaboratively and generate a comprehensive list of threats and opportunities.
-
Checklists – Various Types — Understand how structured checklists can help ensure consistency and completeness in identifying known risk factors and compliance issues.
-
FMEA – Failure Mode and Effects Analysis (General) — Explore how FMEA identifies potential failure modes in systems, evaluates their impacts, and prioritizes actions to mitigate risk.
-
dFMEA – Design FMEA — Study how Design FMEA is used during product or system design stages to anticipate and address design-related failures before implementation.
-
pFMEA – Process FMEA — Learn how Process FMEA applies to manufacturing or operational processes to identify weak points and prevent quality and performance failures.
-
uFMEA – User FMEA — Understand how User FMEA focuses on product use and human interaction to ensure safety, reliability, and usability from an end-user perspective.
-
FMECA – Failure Mode, Effects and Criticality Analysis — Gain insights into evaluating the criticality of potential failures, combining FMEA with quantitative risk assessment techniques.
-
FTA – Fault Tree Analysis — Learn to apply FTA for analyzing complex systems, identifying root causes of system failures, and mapping logical pathways of fault events.
-
HACCP – Hazard Analysis and Critical Control Points — Understand how HACCP principles identify and manage food safety hazards at critical control points in production.
-
HAZOP – Hazard and Operability Study — Study how HAZOP systematically examines processes to identify deviations from intended operations and their potential safety or environmental impacts.
-
Pareto Charts – The 80/20 Rule — Explore how Pareto analysis helps prioritize risk control actions by focusing on the few causes that contribute most to potential problems.
-
Risk Register — Learn how to document, track, and monitor risks using a risk register, ensuring clear visibility and accountability for mitigation actions.
-
Risk Matrix – Consequence/Likelihood Matrix — Understand how to apply a risk matrix to evaluate risks based on their likelihood and consequence, enabling consistent risk ranking.
-
SWIFT – Structured What-If Analysis — Learn how SWIFT facilitates creative risk identification by posing structured “what-if” questions to reveal potential failures or threats.
-
What materials are included in this ISO 31010 Risk Management Foundation Course?
These include examples. illustrative figures and a set of Hazard Analysis Templates, namely:
- Risk Management Process
- Terms and Definitions for ISO 14971
- Example Design FMEA
- Example Process FMEA
- Example User FMEA
- Example FMECA Rating Table
- Note: Choice of Tools used for Riak Assessment
- Note: HACCP Principles and Hazard Analysis
- Note: Supply Chain Integrity Best Practice
- Fig 1: Hazards – Sequence of Events – Hazardous Situations
- Fig 2: Example of Risk Chart
- Fig 3: Example of Qualitative 3 x 3 Risk Matrix
- Fig 4: Example of Semi-quantitative 5 x 5 Risk Matrix
- Fig 5: Sequence of events and risk – Safety
- Fig 6: Sequence of events and risk – Information Security
- Fig 7: Production and Post-production Information Sources
- Fig 8: Example of the application of the P1 x P2 Approach
- Form – SWIFT What-If Analysis
- Form – Hazard Identification Checklist
- Form – Preliminary Hazard Analysis
- Form – Event Tree Analysis with probabilities
- Form – FMEA Team Start
- Form – Design FMEA Scope Worksheet
- Form – Process FMEA Scope Worksheet
- Form – FMEA Worksheet (Word file)
- Form – FMEA Worksheet (Excel file)
- Example HACCP Plan
How is this ISO 31010 Risk Management Foundation Course delivered?
deGRANDSON’s risk management courses are delivered online from our Learning Management System (LMS) at www.degrandsonLMS.com (provided by Inquisiq).
All Modules have a full resume and scaling capabilities. This means, for example, you can…
- Start a Module at work on your Work Station running on Windows 11,
- Continue the Module on the train home on your iPad running on iOS 16 and,
- Complete the Module at home on your Notebook PC running on Windows 10
Are there any prequalifications to enroll in this ISO 31010 Risk Management Foundation Course?
For this course, the minimum of a Secondary School Certificate (such as a High School Diploma, Baccalaureate or similar National Vocational Qualification) combined with 5 years work experience, with at least 2 at managerial/supervisory level, is recommended.
A working knowledge of ISO 13485 and relevant regulations is needed to get the maximum benefit from this Course.
You do not have to provide us with any evidence of your qualifications and experience. However, if you do not meet these requirements, you can expect to struggle with the Program.
Related Courses
Newsletter Subscription
Get updates on the latest news about ISO management systems or the latest promotional offers.
Course Author

Dr John FitzGerald graduated with a 1st class honours degree in chemistry and a PhD in synthetic organic chemistry. He worked for 15 years in the manufacturing industry, then as a trainer and consultant in the UK and Ireland before founding deGRANDSON Global in 2009.
He serves as the company Director and course developer while occasionally working as a Lead Auditor on ISO 9001, ISO 13485, ISO 14001, ISO 27001, ISO 45001, and ISO 55001 audits for an accredited certification body (CAB).
Our Certification

deGRANDSON Global has three university-grade management system certification including ISO 21001, Educational Organizational Management System; ISO 29993, Learning Services outside formal Education; and ISO 29994, Learning Services – additional requirements for Distance Learning.
Course Finder Tool
Not sure if this is the right course for you? Use our Course Finder tool to find out which course will suit your needs best or book a call with the Director and course developer for personal advice.
Get Answers to Frequently Asked Questions
See answers to questions about course content, course delivery, enrollment process, registration and payment, technical requirements and troubleshooting, certification exam and issuance of certificates, free ISO standards, and learner support
Free Sample Lesson
Get a preview of the deGRANDSON e-Training Method for yourself with our free 5-minute sample lesson or get the full immersive experience with our free 30-minute sample lesson.
Sample Learner Certificate

deGRANDSON Global certificates issued to learners who successfully completed the training and passed the certification exam come with QR codes that can be shared on online profiles to instantly verify qualification to prospective clients or employers.