ISO 13485:2016 Transition Training Course
Need to undertake an ISO 13485 2016 Migration?
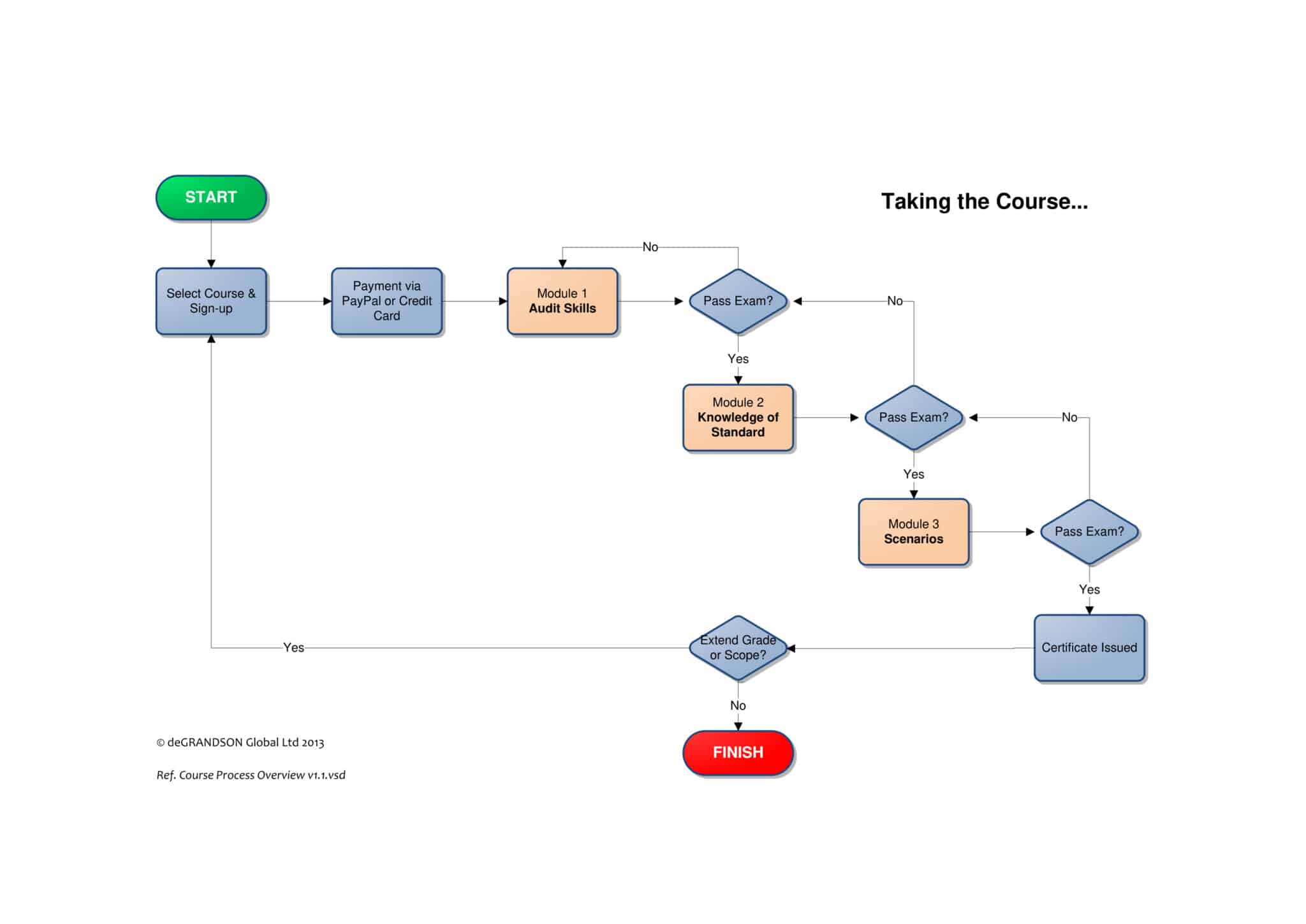
Transitioning to ISO 13485:2016 is a challenge that Quality Managers and others responsible for their organization's Medical Device Management System (MDMS) will face shortly. When you're ready to make the change, this ISO 1345 transition training course will take you step-by-step through the transition process and enable your organization to meet all of the requirements of ISO 13485:2016. In addition, this program will provide you personally with Lead Implementer & Auditor Certification.
Key Features:
- Course is accredited to ISO 21001, the standard for Educational Organizations Management Systems
- An ISO 13485 Lead Implementer certification is awarded upon passing the certification exam
- Certificate is immediately available online after the successful passing of the exam
- Certificate comes with a shareable QR code for instant verification of credentials
- Lessons range from 15 minutes to 1 hour, typically 20–30 minutes, ensuring that each topic is covered in suitable detail
- Course includes practice with scenarios that include dialogues
- Course includes 24/7 Live-chat Learner Support
- Course includes a learner manual, a copy of the standard, and samples of relevant forms and other documents
- Course comes with full-audio narration and Closed Captions for accessibility
- Courses are hosted on your browser so that no software has to be downloaded avoiding security risks.
- Course comes with quizzes, practice with scenarios, and open-book certification exam
- Course is hosted on your browser so that no software has to be downloaded avoiding security risks.
- Features cross-device compatibility (courses can be taken on any desktop, tablet, or mobile)
- Offers full-resume feature (end a session mid-lesson and continue exactly where you left off, even from a different device)
- Features real-time interactive content in a secure web-based environment
- Offers a clear learning path (once you've completed the internal auditor course, you have the option to progress to the lead auditor, up to the consultant and lead auditor course.
- Examination and certificate fee are already included in the course fee
- Payable via PayPal or Stripe using any credit or debit cards
- Option to pay in 4 monthly installments available
Who should enroll in this ISO 13485 Transition Training?
This ISO 13485 Transition Training Course is intended for:- Senior quality managers
- Quality professionals and consultants
- Regulatory professionals
- Compliance professionals
- Project managers
- Design engineers
- Software engineers
- Process owners
- Quality engineers
- Quality auditors
- Start a Module at work on your Work Station running on Windows 11,
- Continue the Module on the train home on your iPad running on iOS 15 and,
- Complete the Module at home on your Notebook PC running on Windows 10.
Are there any prequalifications to enroll in this ISO 13485 Transition Training?
If you have previously completed a Lead Implementer or Lead Auditor Program with deGRANDSON Global, you are pre-qualified. Just log-in, enroll and start your program immediately.
If you have NOT previously completed an Internal Auditor Program with deGRANDSON Global, you are advised to complete a free Pre-Test first on order to demonstrate adequate auditing skills. The Pre-Test will take 30 minutes approx. and is FREE. Click this link to begin: Check your Lead Auditor Skills.
Regarding prerequisite qualifications generally for this, the minimum of a Secondary School Certificate (such as a High School Diploma, Baccalaureate or similar National Vocational Qualification) combined with 5 years’ work experience, with 2 at managerial/supervisory level, is recommended. You do not have to provide us with any evidence of your qualifications and experience. However, if you do not meet these requirements, you can expect to struggle with this program.